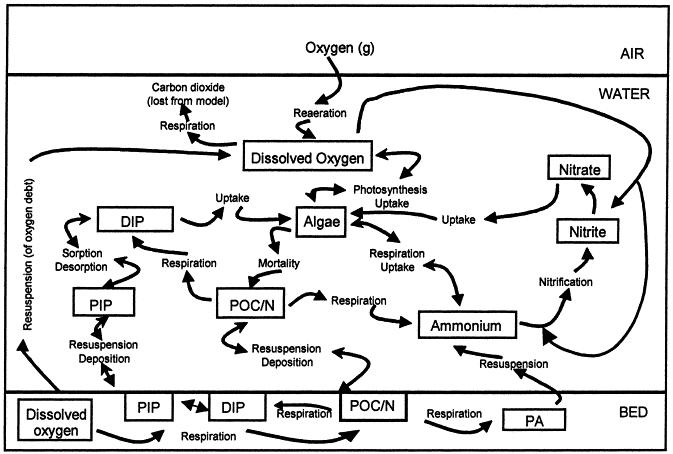
Chemical Reactions Involved
Phosphorous exists in two major forms on Earth: white phosphorous and red phosphorous. Because of its high reactivity, phosphorous is never found alone, making it a diatomic element. When exposed to damp air, white phosphorous reacts by glowing in the dark (chemuluminescence). This must be handled with extreme care, as it spontaneously ignites in air. Phosphorous reacts with halogens vigorously at room temperature to form phosphorous trihalides.
Phosphorus reactions include:
Phosphorus reactions include:
- substitution reactions - 1 group of phosphorous is replaced by another group
- oxidation reactions - oxidation state of phosphorous increases after the reaction
- addition reactions - another molecule adds to the phosphorous
- elimination - 1 molecule leads to 2 molecules
- oxidative - combines oxidation and addition